Lithium-ion battery packs used in micro-mobility devices, such as e-bikes and e-scooters can lead to substantial safety hazards should a single cell go into thermal runaway. In this paper we explore the extent and severity of e-mobility battery fires resulting from a single cell thermal runaway failure and evaluate various suppression techniques a user may attempt to implement if they experience a battery fire at home. We tested a household water hose as well as different fire blankets deployed both before the forced thermal runaway event and after initiation. The water hose was unable to supply a sufficient amount of water to extinguish the thermal event, however, the average pack temperature was decreased and the cell-to-cell propagation rate was slowed. Neither fire blanket tested was able to contain the flames or debris ejected from the battery packs and both acted to hold in the heat from the event, increasing the temperature, rather than allowing it to dissipate. In addition, we also demonstrated how various design approaches, such as added thermal insulation between cells, can help prevent cell-to-cell propagation and reduce the severity of a battery pack failure.

The Electrochemical Society (ECS) was founded in 1902 to advance the theory and practice at the forefront of electrochemical and solid state science and technology, and allied subjects.
ISSN: 2754-2734
ECS Advances is a gold open access journal covering all technical areas supported by the Electrochemical Society. Free for readers and researchers.
Why should you publish in ECS Advances?
ECS Advances carries the broadest dissemination of electrochemical and solid-state science & technology content among all journals in the field, coupled with a rigorous peer review.
Open access: Enabling open access to scientific research is imperative to ECS in our mission to disseminate the best research in our technical fields as widely as possible.
In the spirit of maintaining the ECS standard of excellence and quality scholarly publications, all papers submitted to ECS Advances will be rigorously peer-reviewed before acceptance.
The editorial board of ECS Advances is a combination of the cumulative editorial teams of The Journal of the Electrochemical Society (JES) and the ECS Journal of Solid State Science and Technology (JSS), charged with continuing the standard of excellence for ECS publications. Editorial board members are leaders in the field, vetted by the Editors-in-Chief, and leading committees of the ECS, an organization with a 120+ year legacy of excellence.
Open all abstracts, in this tab
Daniel A. Torelli et al 2024 ECS Adv. 3 010501
Weiran Zheng 2023 ECS Adv. 2 040502
Python, an open-source, interpreted programming language, has emerged as a transformative force within the scientific community, captivating researchers with its rich ecosystem of packages and syntax that prioritizes readability and simplicity. In the rapidly evolving field of electrochemistry, where the analysis of complex data sets, custom analysis routines, and theoretical simulations are indispensable, Python's capabilities have garnered significant attention. This review serves as a general introduction to the utilization of Python in electrochemistry, focusing on beginners who are new to programming concepts.
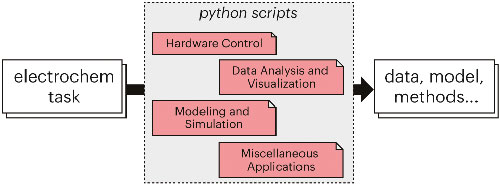
Alexander Frank et al 2022 ECS Adv. 1 040502
The 18 650 and 21 700 cell format are state of the art for high-energy cylindrical lithium-ion batteries, while Tesla proposed the new 4680 format with a continuous "tabless" design as the choice for electric vehicle applications. Using an experimentally validated multidimensional multiphysics model describing a high energy NMC811/Si-C cylindrical lithium-ion battery, the effects of tabless design and cooling topologies are evaluated for 18 650, 21 700, and 4680 cell formats under varying charging protocols. Mantle cooling is found to be the most efficient cooling topology for a segmented tab design, whereas tab cooling performs equally well for tabless cells and achieves better performance for the 4680 format. By massively reducing polarization drops (approx. 250 mV at 3C) and heat generation inside the current collectors (up to 99%), the tabless design increases cell homogeneity and enables format-independent scalability of fast-charging performance with a tab-cooling topology. In addition, the 0 to 0.8 SoC charge time can be reduced by 4 to 10 min compared to cells with a segmented tab design, resulting in 16.2 min for the 18 650 and 21 700, and 16.5 min for the larger 4680 cell format.
Xin Gao et al 2024 ECS Adv. 3 024501
Addressing climate change by carbon management is critical to achieving the goal of net zero carbon emissions by 2050. In this work, we examined the electrochemically-driven recovery of CO2 during alkaline solvent regeneration for solvent-based direct air capture. A mathematical model was developed by incorporating carbonate chemistry with water electrolysis to predict the energy consumption per unit of CO2 released. The predicted results were consistent with the experimental data, in which the experimental work was achieved by characterizing alkalinity and carbon loading values of solvent collected from a flow carbonate electrolyzer. Through this study, we learned that minimizing the energy expended on CO2 release can be achieved by using an anolyte with a lower alkalinity, increasing the electric charge input to the electrolyzer, and reducing the ohmic resistance of the electrolyzer. Furthermore, using a supporting electrolyte, e.g., Na2SO4 in the present work, effectively compensates for the higher ohmic resistance from using an anolyte with a lower alkalinity.
Zhuoming Feng et al 2024 ECS Adv. 3 024502
The effect of an Al2TiO5 (ALT) interlayer between Ni and YSZ on enhancing the thermal stability of Ni-YSZ solid oxide fuel cell was examined. Atomic layer deposition (ALD) was used to provide precise control of the structure and thickness of the ALT interlayer. The study's findings demonstrate that a 2 nm thick ALT interlayer deposited by ALD does not adversely affect the cell's ohmic resistance and effectively prevents Ni sintering and the loss of active area during high-temperature heat treatments. ALT layers thicker than 2 nm, although they enhanced Ni stability, were found to impede oxygen ion transport in the electrode and significantly increase the ohmic resistance of the cell, leading to a decline in performance.

Andrei Kulikovsky 2024 ECS Adv. 3 024503
Recently, it was shown that performance of the cathode catalyst layer in PEM fuel cell dramatically improves if the oxygen concentration (OC) at the catalyst/ gas diffusion layer (GDL) interface oscillates in–phase with the overpotential. However, OC oscillations could only be organized in the cathode channel. Does transport through the GDL affect the phase angle of OC oscillations? Below, it is shown that at low frequencies below 1 Hz, the GDL does not change this phase angle. At high frequencies, the phase angle after transport through the GDL is −3π/4 regardless of the GDL parameters.

Highlights
Analysis of the phase shift of the oxygen concentration oscillations upon transport through the GDL is performed.
At frequencies below 1 Hz, the phase shift due to GDL is zero.
Oxygen concentration in cathode channel oscillating in-phase with the overpotential greatly improves cell performance.
Youyi Sun 2024 ECS Adv. 3 010503
In the research for the catalytic activity of metals, the electronegativity (EN) and valence electronic configuration (VEC) of the atoms of metals have been considered as important factors. By comparing the catalytic activity of the hydrogen evolution reaction (HER) from various metals, we find that metals with high EN and containing two or more accessible partially filled orbitals (PFOs) can support high exchange current density i0,H(A cm−2) for HER, however, metals with low EN and/or with just one or no PFOs are unable to support high i0,H values. It is, therefore, concluded that the EN and the PFOs of the atoms of metals plays a decisive role in its catalytic behavior. Here, the EN and the VEC of the atoms of transition metals in relation to the catalytic activation for HER is discussed and a new type of reaction mechanism of HER on various metal electrodes is also suggested.
Peng He et al 2024 ECS Adv. 3 010504
In-situ polymerization is a method for fabricating lithium-ion batteries to contain solid or gel electrolytes without major manufacturing changes. Gel polymer electrolytes (GPEs) wherein some polymer in incorporated, decreasing the volume of liquid electrolyte, have been pursued as they may be safer. One challenge with GPEs is reduced ion transport properties. In this work, macromonomers with different chain chemistry and ionic monomers are crosslinked on electrospun polyvinylidene difluoride (PVDF) in the presence of solvents, without conventional salt, to in-situ fabricate GPEs with elevated transference number within coin cells. These GPEs would be single-ion conductors in the case of complete ionic monomer polymerization to the crosslinked network. The effect of chain chemistry and the percentage of macromonomers and ionic monomers in the GPEs on conductivity are investigated. It is found that poly(siloxane) diacrylate (PDMSDA)- and perfluoropolyether tetra-acrylate (PFPETA)-based GPEs outperform the poly(propylene glycol) diacrylate (PPGDA)- and poly(tetrahydrofuran) diacrylate (PTHFDA)-based GPEs in terms of ionic conductivity. The highest ionic conductivity was achieved for a PDMSDA-based GPE at 4.2 × 10−4 S cm−1 at 23 °C. Graphite/NMC-811 full cells prepared with the in-situ polymerized PDMSDA-based GPEs show capacity retention of 82.6% after 100 cycles, albeit with limited electrode utilization due to ion transport limitations.
A. N. Payzullaev et al 2023 ECS Adv. 2 031001
This study investigated the impact of silicon dioxide nanoparticle size on the viscosity and stability of nanofluids. A comprehensive study was conducted, including the synthesis and characterization of silicon dioxide nanoparticles of varying sizes, as well as the preparation and testing of nanofluids with different nanoparticle concentrations. The results show that nanoparticle size has a significant effect on both the viscosity and stability of nanofluids, with smaller nanoparticles leading to higher viscosity and improved stability. Silicon dioxide nanoparticles were dispersed in deionized water at mass fractions of 0.5% and 3% and average particle diameters of 12 nm and 40 nm using an ultrasonic disperser. The rheological properties of the obtained nanofluids at 304 K were studied depending on the mass concentration and linear size of nanoparticles in the liquid. An analysis of the results showed that the viscosity of the nanofluids increased with decreasing linear size of the nanoparticles. These findings have important implications for the development and optimization of nanofluid-based applications in various industries, such as electronics, energy, and biomedical engineering.
Francielli Silva Genier et al 2022 ECS Adv. 1 030502
Deriving battery grade materials from natural sources is a key element to establishing sustainable energy storage technologies. In this work, we present the use of avocado peels as a sustainable source for conversion into hard carbon-based anodes for sodium ion batteries. The avocado peels are simply washed and dried then proceeded to a high temperature conversion step. Materials characterization reveals conversion of the avocado peels in high purity, highly porous hard carbon powders. When prepared as anode materials they show to the capability to reversibly store and release sodium ions. The hard carbon-based electrodes exhibit excellent cycling performance, namely, a reversible capacity of 352.55 mAh g−1 at 0.05 A g−1, rate capability up to 86 mAh g−1 at 3500 mA g−1, capacity retention of >90%, and 99.9% coulombic efficiencies after 500 cycles. Cyclic voltammetry studies indicated that the storage process was diffusion-limited, with diffusion coefficient of 8.62 × 10−8 cm2 s−1. This study demonstrates avocado derived hard carbon as a sustainable source that can provide excellent electrochemical and battery performance as anodes in sodium ion batteries.
Open all abstracts, in this tab
Weiran Zheng 2023 ECS Adv. 2 040502
Python, an open-source, interpreted programming language, has emerged as a transformative force within the scientific community, captivating researchers with its rich ecosystem of packages and syntax that prioritizes readability and simplicity. In the rapidly evolving field of electrochemistry, where the analysis of complex data sets, custom analysis routines, and theoretical simulations are indispensable, Python's capabilities have garnered significant attention. This review serves as a general introduction to the utilization of Python in electrochemistry, focusing on beginners who are new to programming concepts.

M. Susaritha 2023 ECS Adv. 2 031003
Various interconnects utilised in very large-scale integration in this work. The expanding use of portable devices has increased the demand for low-power circuit design. Sub-threshold circuits are the greatest option to address the demand for even more-low power. However, decreased performance and increased variability are the main problems with sub-threshold circuits. Furthermore, global interconnects have a significant impact on the performance and power dissipation of sub-threshold circuits. For future VLSI circuit applications, interconnect is a brand-new and very promising alternative that has to have its propagation latency and stability analysed in order to substantiate its claim that it can replace existing interconnect designs.
Hamidreza Parsimehr et al 2023 ECS Adv. 2 020503
Energy production/storage as one of the world's leading issues has been widely studied. This significant problem can be solved by using disposable/low-cost biomass materials. Electrochemical energy storage (EES) devices including supercapacitors, rechargeable batteries, and hybrid EES devices have been extensively developed in recent years. The EES devices have been recognized as a proper answer to the energy storage problem in the world. Numerous studies have been accomplished to develop biomass-based and biochar-based EES devices to decrease environmental pollution and production costs. The most important part of the EES devices are electrodes including the cathode and anode. According to recent studies, biochar-based electrodes have considerable electrochemical properties. Wheat is one of the most important parts of the human diet. The wheat wastes have amazing electrochemical properties to be used as a precursor of electrochemical energy storage (EES) electrodes including supercapacitors, batteries, and hybrid EES devices. The benign/low-cost wheat wastes especially wheat straw and wheat husk have been used to fabricate wheat-based biochar materials. The electrochemical properties of the wheat-based biochar electrodes (cathode and anode) in the EES devices have determined that these benign/low-cost EES electrodes reduce production costs and obtain acceptable electrochemical performance and environmentally friendly procedures.
Dola Sundeep et al 2023 ECS Adv. 2 016502
Covid-19 still remains as global pandemic surging with different variants like alpha, beta, gamma, delta and most omicronin various countries resulted into SARS-CoV-2. An escalation in random testing for Covid-19 is considered as golden standard by World Health Organization (WHO) to control the spread of Corona virus. RT-PCR and nucleic acid hybridization strategies are the two highly sensitive outstanding tests used for the detection of SARS-CoV-2 in the DNA by the former and RNA/DNA by the later. Apart from the test for virus, antigen and antibodies other alternative hematological tests like CRP, Ferritine, IL-6, Blood Platelets and WBC and plasma became essential for the detection and management of Covid-19. Overcoming the limitations a rapid, efficient, low-cost and easy-to-implement test kits are in imperative need to meet the requirement. Electrochemical sensors are considered as potential, ultra-fast and point-of-care sensors used as self-testing kits. In this work, a meta review of various modified electrodes with different nanomaterials such as graphene, CNTs, and nano-dopants used as electrochemical sensors in association with testing various different parameters for detection of Covid-19. A detailed discussion on the merits and demerits of present testing patterns and scope of screen-printed electrochemical sensors modified with different nanomaterials and their advantages are presented.

Bongiwe Silwana and Mangaka C. Matoetoe 2022 ECS Adv. 1 046502
Since the 1980s, the HIV/AIDS pandemic has been a global concern and remains despite the tremendous efforts and successes in the development of antiviral drugs. The drug development and formulation process are not without challenges. However, the effective use of excipients can allow formulators to circumvent or at least minimize the impact of these challenges. Some of the challenges include ensuring a high quality of actives, free from impurities, and ensuring administration in appropriate quantities. Incorrect dosing can have a detrimental impact on the body. Evaluation of the quality and physiological function of these drugs in pharmaceutical and biological samples is vital. Considering this, we explore the potential application of electrochemical methods for the detection of these drugs. Electrochemical methods are versatile and enable faster and cheaper detection in comparison to conventional techniques. This review paper provides an outline of the latest developments and new trends in electroanalysis of the first generation of nonnucleoside reverse transcriptase inhibitors class of drugs. Furthermore, we highlight several phenomena occurring on bare solid electrodes and modified electrodes, including underlying mechanisms. In addition, we investigate the physical and electrochemical properties together with their figures of merits. The review covers only the reactions occurring at the surface of bare and modified electrodes and the reader is referred to the cited literature for the corresponding voltammograms of these studies. Recent data shows that modified electrodes based on nanoparticles present substantial advantages over their non-modified counterparts enabling portability, higher selectivity, and multiple detection capabilities. These characteristics make nanoparticles promising candidates for the design of sensors.

Open all abstracts, in this tab
Nguyen et al
The influence of different synthesis parameters on CeO2 and Pt nanoparticle (NP) deposition on Ketjenblack carbon (C(KB)) was examined. The Pt NP diameter (3.1–4.1 nm) was not influenced by CeO2 synthesis parameters. The CeO2 NPs synthesized using ultrasound sonication contribute to a better durability of the Pt-CeO2/C against CO poisoning. In contrast, CeO2 synthesized using the microwave heating method contributes to better methanol oxidation reaction (MOR) activity at low potential. Synthesis parameters of CeO2 and Pt NPs developed for the C(KB)-based catalysts were applied for C(Cr3C2)-based catalysts. The Pt NP diameter of C(Cr3C2)-based catalysts was slightly higher (7.2 nm) as some Pt NPs were agglomerated. The C(Cr3C2) support facilitates the MOR and CO stripping, especially in the case of the Pt/C on C(Cr3C2) support. The MOR activity at 0.85 V of Pt/C on the C(Cr3C2) support is as good as the MOR activity for the best Pt-CeO2/C on the C(KB) support. The C(Cr3C2) support also improves the CO removal from the Pt surface. All the synthesized catalysts had better MOR activity than the commercial Pt/C(Vulcan) catalyst. The oxygen reduction reaction activity of Pt-CeO2/C catalysts with higher CeO2 content synthesized with the microwave heating method was very good.