Journal Description
Molbank
Molbank
is an international, peer-reviewed, open access journal comprised of a unique collection of one-compound-per-paper short notes on synthetic compounds and natural products published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), Reaxys, CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 15.5 days after submission; acceptance to publication is undertaken in 2.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
0.6 (2022)
Latest Articles
Adamantylated Calix[4]arenes Bearing CuAAC-Ready 2-Azidoethyl or Propargyl Functionalities
Molbank 2024, 2024(2), M1821; https://doi.org/10.3390/M1821 (registering DOI) - 11 May 2024
Abstract
1,3-Alternate calix[4]arenes were prepared, having bulky adamantyl groups in the p-positions of all four aromatic units of the macrocycles and pairs of propargyl or 2-azidoethyl groups alternating with n-propyl groups at the phenol oxygen atoms. The step-wise syntheses were carried out
[...] Read more.
1,3-Alternate calix[4]arenes were prepared, having bulky adamantyl groups in the p-positions of all four aromatic units of the macrocycles and pairs of propargyl or 2-azidoethyl groups alternating with n-propyl groups at the phenol oxygen atoms. The step-wise syntheses were carried out through a selective distal alkylation of the parent p-adamantylcalix[4]arene with propargyl bromide or 1,2-dibromoethane, resulting in calix[4]arenes bearing pairs of propargyl or 2-bromoethyl groups at their narrow rims. The bromine atoms were replaced by azide groups, and then both calix[4]arene diethers were exhaustively alkylated at the remaining OH-groups with 1-iodopropane under stereoselective conditions to fix the macrocycles in an 1,3-alternate shape. The structures of the prepared p-adamantylcalix[4]arenes were confirmed by NMR and HRMS data, and, for the 1,3-alternate dipropargyl ether, the X-ray diffraction data were also collected. Preliminary data on the reactivity of the prepared calixarenes under the CuAAC conditions suggested a strong steric hampering created by the adamantane units nearby the reacting alkyne or azide groups that affected the outcome of the two-fold cycloaddition involving the calixarene bis(azides) or bis(alkynes) as complementary partners.
Full article
(This article belongs to the Section Organic Synthesis)
►
Show Figures
Open AccessShort Note
8(S)-[(1R,2S,5R)-2-Isopropyl-5-methylcyclohexyloxy]-3,4-dihydro-2H-[1,4]dithiepino[2,3-c]furan-6(8H)-one
by
Alsu M. Khabibrakhmanova, Enze S. Rabbanieva, Darya P. Gerasimova, Liliya Z. Latypova and Almira R. Kurbangalieva
Molbank 2024, 2024(2), M1820; https://doi.org/10.3390/M1820 (registering DOI) - 11 May 2024
Abstract
The interaction of propane-1,3-dithiol with the chiral bis-thioether, which combines two 2(5H)-furanone moieties, bridged through their carbon atoms C(4) by the propane-1,3-dithiol fragment, in DMF in the presence of potassium hydroxide or cesium carbonate resulted in the formation of an
[...] Read more.
The interaction of propane-1,3-dithiol with the chiral bis-thioether, which combines two 2(5H)-furanone moieties, bridged through their carbon atoms C(4) by the propane-1,3-dithiol fragment, in DMF in the presence of potassium hydroxide or cesium carbonate resulted in the formation of an optically active fused bicyclic sulfur heterocycle, possessing 1,4-dithiepine and unsaturated γ-lactone moieties. The studied reaction follows an unexpected pathway in a basic medium with the thiolate–thiolate exchange. The structure of the novel heterocycle of the 1,4-dithiepinofuranone series is characterized by single-crystal X-ray diffraction.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures

Figure 1
Open AccessCommunication
Synthesis and Structure of a Coordination Polymer of Ni(II) with 2-(4-Bromophenoxy)acetohydrazide
by
Оlena Martsynko, Mariia Nesterkina, Оlena Finik, Kyrylo Tsymbaliuk, Viktoriya Dyakonenko, Svitlana Shishkina and Iryna Kravchenko
Molbank 2024, 2024(2), M1819; https://doi.org/10.3390/M1819 - 8 May 2024
Abstract
►▼
Show Figures
The coordination compound [NiCl2L(2-PrOH)]n (where L is 2-(4-bromophenoxy)acetohydrazide; 2-PrOH is isopropanol) was obtained for the first time. The complex was characterized by means of elemental analyses, molar conductance, thermogravimetric analysis, IR spectroscopy, and single crystal X-ray diffraction analysis. It was
[...] Read more.
The coordination compound [NiCl2L(2-PrOH)]n (where L is 2-(4-bromophenoxy)acetohydrazide; 2-PrOH is isopropanol) was obtained for the first time. The complex was characterized by means of elemental analyses, molar conductance, thermogravimetric analysis, IR spectroscopy, and single crystal X-ray diffraction analysis. It was determined that the coordination compound exhibits a polymeric structure. The complexing agent is six-coordinated, and its distorted octahedron forms due to the bidentate coordination of 2-(4-bromophenoxy)acetohydrazide through the carbonyl oxygen atom and the amine nitrogen. The oxygen of the molecule of isopropanol, the chlorine atom, and two chlorine atoms serve as bridges between two metal atoms.
Full article
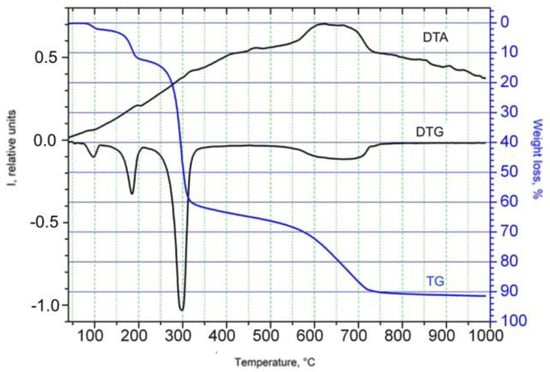
Figure 1
Open AccessShort Note
(E)-1-(5-(Hydroxymethyl) furan-2-yl)-4,4-dimethylpent-1-en-3-one
by
Zhongwei Wang, Luxiao Zhou, Peng He and Yukun Qin
Molbank 2024, 2024(2), M1818; https://doi.org/10.3390/M1818 - 7 May 2024
Abstract
►▼
Show Figures
This study presents a novel approach in the realm of catalytic organic synthesis by integrating biomass catalytic conversion with organic synthesis techniques. Utilizing N-acetylglucosamine as the primary feedstock, the first phase of the research involves its catalytic transformation into 5-hydroxymethylfurfural (HMF). The
[...] Read more.
This study presents a novel approach in the realm of catalytic organic synthesis by integrating biomass catalytic conversion with organic synthesis techniques. Utilizing N-acetylglucosamine as the primary feedstock, the first phase of the research involves its catalytic transformation into 5-hydroxymethylfurfural (HMF). The subsequent phase employs a condensation reaction between HMF and 3,3-Dimethyl-2-butanone to synthesize a new compound, (E)-1-(5-(hydroxymethyl) furan-2-yl)-4,4-dimethylpent-1-en-3-one. This two-step process not only demonstrates the feasibility of converting biomass into valuable chemical precursors but also exemplifies the synthesis of novel compounds through green chemistry principles. The successful execution of this methodology offers fresh insights and opens new avenues for advancements in catalytic organic synthesis, emphasizing sustainability and efficiency.
Full article
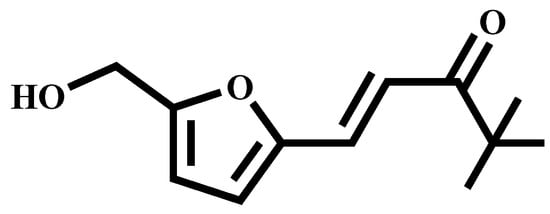
Figure 1
Open AccessShort Note
4,4’-(Thiophene-2,5-diylbis(ethyne-2,1-diyl))bis(1-methyl-1-pyridinium) Iodide
by
Lorenza Romagnoli, Alessandro Latini and Andrea D’Annibale
Molbank 2024, 2024(2), M1817; https://doi.org/10.3390/M1817 - 6 May 2024
Abstract
In the vast field of organic functional materials, viologens are widely recognized as an extremely versatile family of substances, due in part to the possibility of extending conjugation between the terminal pyridinium rings, for instance through the insertion of additional aromatic moieties. In
[...] Read more.
In the vast field of organic functional materials, viologens are widely recognized as an extremely versatile family of substances, due in part to the possibility of extending conjugation between the terminal pyridinium rings, for instance through the insertion of additional aromatic moieties. In this work, a new, extended viologen with a thiophene core and two acetylene bonds is presented. It was synthesized through a straightforward route, using well-established Sonogashira coupling reactions, and its optical properties were investigated by UV–visible absorption and fluorescence spectroscopy, revealing a very interesting material for diverse fluorescence-related applications.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Figure 1
Open AccessShort Note
Di-µ-(1-(3-(1H-imidazol-1-yl)propyl)-2-methyl-4-oxo-1,4-dihydropyridin-3-olate)-bis[(η5-pentamethylcyclopentadienyl)iridium(III)] Chloride
by
Ilya A. Shutkov, Nikolai A. Melnichuk, Konstantin A. Lyssenko, Nataliya E. Borisova, Olga N. Kovaleva and Alexey A. Nazarov
Molbank 2024, 2024(2), M1816; https://doi.org/10.3390/M1816 - 5 May 2024
Abstract
A metallacyclic maltol-tethered organometallic Ir(III) half-sandwich complex was synthesized as an analog of the ruthenium anticancer complexes (RAPTA/RAED) to evaluate its in vitro antiproliferative activity against various human cancer cell lines.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Figure 1
Open AccessShort Note
2S-(1RS-benzyloxy-hex-5-enyl)-2,3-dihydro-1,4-benzodioxine
by
Angelica Artasensi and Laura Fumagalli
Molbank 2024, 2024(2), M1812; https://doi.org/10.3390/M1812 - 24 Apr 2024
Abstract
►▼
Show Figures
In medicinal chemistry, the precise configuration of molecules is a crucial determinant of their pharmacological properties. Hence, the introduction of a new chiral center during the synthetic pathway involves the assignment of configuration. Herein we assign, by means of molecular modeling 1H
[...] Read more.
In medicinal chemistry, the precise configuration of molecules is a crucial determinant of their pharmacological properties. Hence, the introduction of a new chiral center during the synthetic pathway involves the assignment of configuration. Herein we assign, by means of molecular modeling 1H and 2D Nuclear Overhauser Effect NMR techniques, the configuration of the two diastereomers 2S-(1R-benzyloxy-hex-5-enyl)-2,3-dihydro-1,4-benzodioxine and 2S-(1S-benzyloxy-hex-5-enyl)-2,3-dihydro-1,4-benzodioxine, which are useful to synthetize analogs of the potent and highly selective dipeptidyl peptidase IV and carbonic anhydrase inhibitor recently published.
Full article

Figure 1
Open AccessShort Note
5,5′-Thiobis(3-bromoisothiazole-4-carbonitrile)
by
Andreas S. Kalogirou and Panayiotis A. Koutentis
Molbank 2024, 2024(2), M1813; https://doi.org/10.3390/M1813 - 24 Apr 2024
Abstract
The reaction of sodium 2,2-dicyanoethene-1,1-bis(thiolate) with bromine (2 equiv.) in CCl4 gave 3,5-dibromoisothiazole-3-carbonitrile and 5,5′-thiobis(3-bromoisothiazole-4-carbonitrile) in 7% and 18% yields, respectively. The latter novel compound was fully characterized.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Figure 1
attachment
Supplementary material:
Supplementary File 1 (ZIP, 660 KiB)
Supplementary File 2 (MOL, 8 KiB)
Supplementary File 3 (INCHI, 3 KiB)
Supplementary File 4 (MOL, 8 KiB)
Supplementary File 5 (MOL, 8 KiB)
Supplementary File 6 (INCHI, 2 KiB)
Supplementary File 7 (MOL, 8 KiB)
Supplementary File 8 (CIF, 2480 KiB)
Supplementary File 1 (ZIP, 660 KiB)
Supplementary File 2 (MOL, 8 KiB)
Supplementary File 3 (INCHI, 3 KiB)
Supplementary File 4 (MOL, 8 KiB)
Supplementary File 5 (MOL, 8 KiB)
Supplementary File 6 (INCHI, 2 KiB)
Supplementary File 7 (MOL, 8 KiB)
Supplementary File 8 (CIF, 2480 KiB)
Open AccessShort Note
5,6-Diphenyl-1,3,4,7-tetra-p-tolyl-1,3,3a,7a-tetrahydropentaleno[1,2-c]furan
by
Shu-Kai Chen, Yi-Hung Liu and Shiuh-Tzung Liu
Molbank 2024, 2024(2), M1814; https://doi.org/10.3390/M1814 - 24 Apr 2024
Abstract
The reaction of (Z)-5-phenyl-1,3-di-p-tolylpent-2-en-4-yn-1-ol (1) with trimethylsilyl chloride in dichloromethane at ambient temperature gave a dimeric ether compound 2 in 30% yield. Subsequently, heating 2 in toluene under refluxing temperature rendered the title compound quantitatively. The structure
[...] Read more.
The reaction of (Z)-5-phenyl-1,3-di-p-tolylpent-2-en-4-yn-1-ol (1) with trimethylsilyl chloride in dichloromethane at ambient temperature gave a dimeric ether compound 2 in 30% yield. Subsequently, heating 2 in toluene under refluxing temperature rendered the title compound quantitatively. The structure of this tricyclic-fused compound was characterized using NMR, mass spectroscopy, and X-ray crystallography. This unique linear tricyclic fused furan framework is reported for the first time.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Graphical abstract
attachment
Supplementary material:
Supplementary File 1 (PDF, 870 KiB)
Supplementary File 2 (MOL, 2 KiB)
Supplementary File 3 (INCHI, 513 B)
Supplementary File 4 (MOL, 2 KiB)
Supplementary File 5 (MOL, 2 KiB)
Supplementary File 6 (INCHI, 702 B)
Supplementary File 7 (MOL, 2 KiB)
Supplementary File 8 (MOL, 2 KiB)
Supplementary File 9 (INCHI, 546 B)
Supplementary File 10 (MOL, 2 KiB)
Supplementary File 1 (PDF, 870 KiB)
Supplementary File 2 (MOL, 2 KiB)
Supplementary File 3 (INCHI, 513 B)
Supplementary File 4 (MOL, 2 KiB)
Supplementary File 5 (MOL, 2 KiB)
Supplementary File 6 (INCHI, 702 B)
Supplementary File 7 (MOL, 2 KiB)
Supplementary File 8 (MOL, 2 KiB)
Supplementary File 9 (INCHI, 546 B)
Supplementary File 10 (MOL, 2 KiB)
Open AccessCommunication
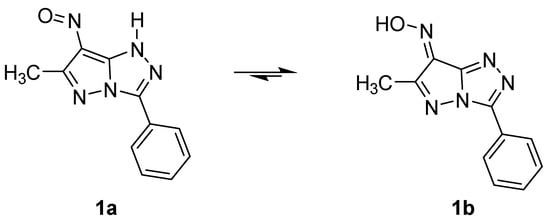
Synthesis and Spectroscopic Study of New 1H-1-Alkyl-6-methyl-7-nitroso-3-phenylpyrazolo[5,1-c][1,2,4]triazoles
by
Ion Burcă, Valentin Badea, Vasile-Nicolae Bercean and Francisc Péter
Molbank 2024, 2024(2), M1815; https://doi.org/10.3390/M1815 - 24 Apr 2024
Abstract
The nitrosation of 1H-1-alkyl-6-methyl-3-phenylpyrazolo[5,1-c][1,2,4]triazoles leads to new 1H-1-alkyl-6-methyl-7-nitroso-3-phenylpyrazolo[5,1-c][1,2,4] triazoles that react in acidic media, giving rise to 1H-1-alkyl-7-hydroxyimino-6-methyl-3-phenylpyrazolo[5,1-c][1,2,4]triazolium salts. These compounds were characterized by FT-IR, UV-Vis, 1H-NMR, 13C-NMR, and 15N-NMR spectroscopic techniques.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures
Scheme 1
Open AccessShort Note
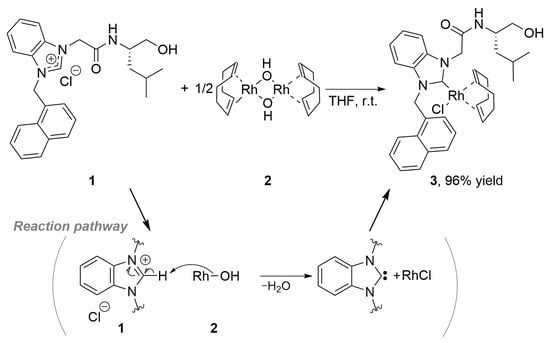
Chloro(η2,η2-cycloocta-1,5-diene){1-[(2-[(S)-1-(hydroxymethyl)-3-methylbutyl]amino)-2-oxoethyl]-3-(1-naphthalenylmethyl)benzimidazol-2-ylidene}rhodium(I)
by
Satoshi Sakaguchi and Shogo Matsuo
Molbank 2024, 2024(2), M1810; https://doi.org/10.3390/M1810 - 19 Apr 2024
Abstract
Commercially available and air- and moisture-stable rhodium complex [Rh(OH)(cod)]2 (2) was utilized in the synthesis of [RhX(cod)(NHC)] (3). The presence of an OH group in complex 2 serves as an internal base, facilitating the deprotonation of the C–H
[...] Read more.
Commercially available and air- and moisture-stable rhodium complex [Rh(OH)(cod)]2 (2) was utilized in the synthesis of [RhX(cod)(NHC)] (3). The presence of an OH group in complex 2 serves as an internal base, facilitating the deprotonation of the C–H bond of the azolium ring in the hydroxyamide-substituted benzimidazolium salt 1. This reaction between 1 and 2 proceeded in THF at room temperature without temperature control, affording the desired NHC/Rh complex 3 in excellent yield. The characterization of complex 3 was accomplished through NMR and HRMS analyses, revealing its existence as a diastereomeric mixture of two NHC/Rh complexes. Furthermore, its catalytic performance was briefly evaluated in the reaction between 2-naphthaldehyde (5) and phenylboronic acid (6).
Full article
(This article belongs to the Topic Heterocyclic Carbene Catalysis)
►▼
Show Figures
Scheme 1
Open AccessShort Note
Chloro(η2,η2-cycloocta-1,5-diene){1-benzyl-3-[(S)-2-hydroxy-1-methylethyl]benzimidazol-2-ylidene}rhodium(I)
by
Satoshi Sakaguchi and Shogo Matsuo
Molbank 2024, 2024(2), M1811; https://doi.org/10.3390/M1811 - 19 Apr 2024
Abstract
Previously, we demonstrated the synthesis of a well-defined hydroxyalkyl-functionalized N-heterocyclic carbene (NHC)/Ru(II) complex through the transmetalation reaction between [RuCl2(p-cymene)]2 and the corresponding NHC/Ag(I) complex derived from a chiral benzimidazolium salt using the Ag2O method. In
[...] Read more.
Previously, we demonstrated the synthesis of a well-defined hydroxyalkyl-functionalized N-heterocyclic carbene (NHC)/Ru(II) complex through the transmetalation reaction between [RuCl2(p-cymene)]2 and the corresponding NHC/Ag(I) complex derived from a chiral benzimidazolium salt using the Ag2O method. In this study, we successfully synthesized [RhX(cod)(NHC)] complexes through a one-pot deprotonation route. The hydroxyalkyl-substituted benzimidazolium salt reacted with [Rh(OH)(cod)]2 in THF at room temperature, affording the corresponding monodentate NHC/Rh(I) complex in nearly quantitative yield. The rhodium complex was characterized using NMR, HRMS measurement, and elemental analysis.
Full article
(This article belongs to the Topic Heterocyclic Carbene Catalysis)
►▼
Show Figures
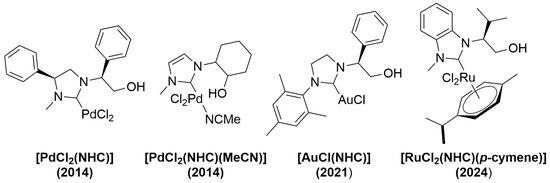
Scheme 1
Open AccessShort Note
Diiodido-bis{N-[2-(diphenylphosphino)benzylidene]benzylamine-κ2N,P}dicopper(I)
by
Julian Süß, Uwe Monkowius and Manfred Zabel
Molbank 2024, 2024(2), M1808; https://doi.org/10.3390/M1808 - 18 Apr 2024
Abstract
The one-pot template reaction between 2-(diphenylphosphino)benzaldehyde, benzylamine and copper(I) iodide yields the dinuclear copper complex (P∩N)2Cu2I2, as revealed by single-crystal X-ray diffraction.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures
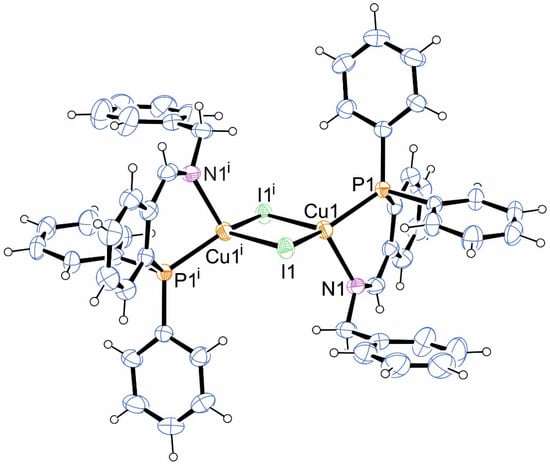
Figure 1
Open AccessShort Note
1-(3-Chlorophenyl)-3-(6-((1,7,7-trimethylbicyclo[2.2.1]heptan-2-ylidene)amino)hexyl)thiourea
by
Daria Zapravdina and Vladimir Burmistrov
Molbank 2024, 2024(2), M1809; https://doi.org/10.3390/M1809 - 18 Apr 2024
Abstract
The compound 1-(3-chlorophenyl)-3-(6-((1,7,7-trimethylbicyclo[2.2.1]heptan-2-ylidene)amino)hexyl)thiourea was synthesized for the first time from 6-((1,7,7-trimethylbicyclo[2.2.1]heptan-2-ylidene)amino)hexan-1-amine and 3-chlorophenylisothiocyanate in DMF with a 60% yield. It was characterized by 1H, 13C{1H} NMR, FT-IR, MS, and elemental analysis.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures
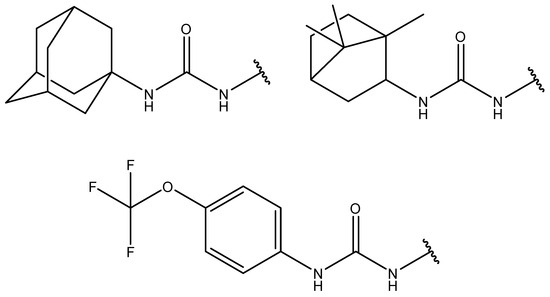
Figure 1
Open AccessShort Note
3,3’-(4,11-Bis(4-(trifluoromethyl)benzyl)-1,4,8,11-Tetraazacyclotetradecane-1,8-diyl)dipropanenitrile
by
Inês M. Nunes, Elisabete R. Silva and Luis G. Alves
Molbank 2024, 2024(2), M1807; https://doi.org/10.3390/M1807 - 17 Apr 2024
Abstract
The cyclam derivative (NCCH2CH2)2(4-CF3PhCH2)2Cyclam was prepared by the reaction of H2(4-CF3PhCH2)2Cyclam with acrylonitrile in methanol. The compound was fully characterized by elemental analysis,
[...] Read more.
The cyclam derivative (NCCH2CH2)2(4-CF3PhCH2)2Cyclam was prepared by the reaction of H2(4-CF3PhCH2)2Cyclam with acrylonitrile in methanol. The compound was fully characterized by elemental analysis, mass spectrometry as well as IR and NMR spectroscopy. Crystals of (NCCH2CH2)2(4-CF3PhCH2)2Cyclam suitable for single-crystal X-ray diffraction were obtained by the slow evaporation of a chloroform solution of the compound. The establishment of non-classical hydrogen bonds and unusual nitrile–nitrile and π(CN)…π interactions determined the solid-state supramolecular architecture of (NCCH2CH2)2(4-CF3PhCH2)2Cyclam.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures
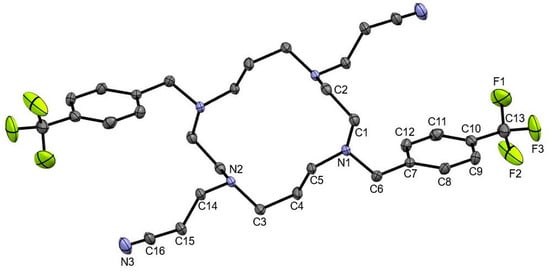
Figure 1
Open AccessCommunication
Synthesis and Analytical Characterization of Cyclization Products of 3-Propargyloxy-5-benzyloxy-benzoic Acid Methyl Ester
by
Matteo Mori, Giulia Cazzaniga, Donatella Nava and Elena Pini
Molbank 2024, 2024(2), M1806; https://doi.org/10.3390/M1806 - 16 Apr 2024
Abstract
In the context of our ongoing studies on chromane derivatives as inhibitors of the salicylate synthase from M. tuberculosis, we isolated a new, unexpected compound from the cyclization of 3-(propargyloxy)-5-benzyloxy-benzoic acid methyl ester. Its molecular structure was elucidated by means of 1D
[...] Read more.
In the context of our ongoing studies on chromane derivatives as inhibitors of the salicylate synthase from M. tuberculosis, we isolated a new, unexpected compound from the cyclization of 3-(propargyloxy)-5-benzyloxy-benzoic acid methyl ester. Its molecular structure was elucidated by means of 1D and 2D NMR analyses, FT-IR, ESI-MS, and HRMS.
Full article
(This article belongs to the Collection Molecules from Side Reactions)
►▼
Show Figures

Figure 1
Open AccessCommunication
The Synthesis and Structure of Scandium Dichloride of Sterically Demanding Aminopyridinato Ligands
by
Sadaf Qayyum
Molbank 2024, 2024(2), M1805; https://doi.org/10.3390/M1805 - 10 Apr 2024
Abstract
The reaction of the potassium salt of (2,6-diisopropylphenyl)-[6-(2,4,6-triisopropylphenyl)-pyridine-2-yl]-amine (Ap*H) with the equimolar ratio of [ScCl3] in tetrahydrofuran (thf) resulted in the mononuclear mono(aminopyridinato) scandium dichloride complex [Ap*ScCl2(thf)2]. An X-ray analysis showed the title compound to be monomeric.
[...] Read more.
The reaction of the potassium salt of (2,6-diisopropylphenyl)-[6-(2,4,6-triisopropylphenyl)-pyridine-2-yl]-amine (Ap*H) with the equimolar ratio of [ScCl3] in tetrahydrofuran (thf) resulted in the mononuclear mono(aminopyridinato) scandium dichloride complex [Ap*ScCl2(thf)2]. An X-ray analysis showed the title compound to be monomeric. The compound [C40H59Cl2N2O2Sc] crystallized in the monoclinic space group, P21/n, and possessed the following cell parameters: a = 12.4441(8) b = 22.9975(10) c = 13.9971(8) Å, β = 92.297(5)°, V = 4002.5(4) A3, and Z = 4. Hirshfeld analyses show that H…H (91.1%), H…C/C…H (5.0%), and H…Cl/Cl…H (3.9%) are the contributing interactions in the solid-state structure. The compound was further characterized by NMR spectroscopy, and its purity was confirmed by elemental analysis.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures
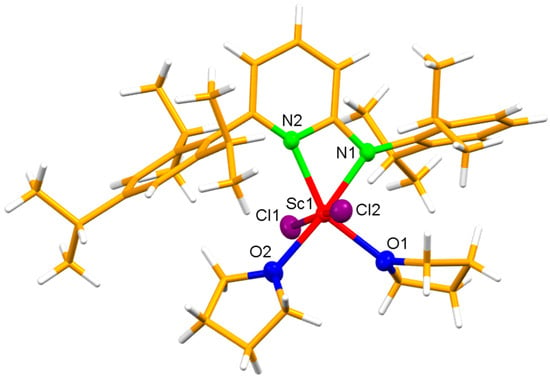
Figure 1
attachment
Supplementary material:
Supplementary File 1 (PDF, 2182 KiB)
Supplementary File 2 (MOL, 2 KiB)
Supplementary File 3 (INCHI, 593 B)
Supplementary File 4 (MOL, 2 KiB)
Supplementary File 5 (MOL, 2 KiB)
Supplementary File 6 (INCHI, 594 B)
Supplementary File 7 (MOL, 2 KiB)
Supplementary File 8 (MOL, 1 KiB)
Supplementary File 9 (INCHI, 365 B)
Supplementary File 10 (MOL, 1 KiB)
Supplementary File 1 (PDF, 2182 KiB)
Supplementary File 2 (MOL, 2 KiB)
Supplementary File 3 (INCHI, 593 B)
Supplementary File 4 (MOL, 2 KiB)
Supplementary File 5 (MOL, 2 KiB)
Supplementary File 6 (INCHI, 594 B)
Supplementary File 7 (MOL, 2 KiB)
Supplementary File 8 (MOL, 1 KiB)
Supplementary File 9 (INCHI, 365 B)
Supplementary File 10 (MOL, 1 KiB)
Open AccessShort Note
1,2-Dibenzoylthiosemicarbazide
by
Burcă Ion, Badea Valentin, Todea Anamaria and Bercean Vasile-Nicolae
Molbank 2024, 2024(2), M1804; https://doi.org/10.3390/M1804 - 9 Apr 2024
Abstract
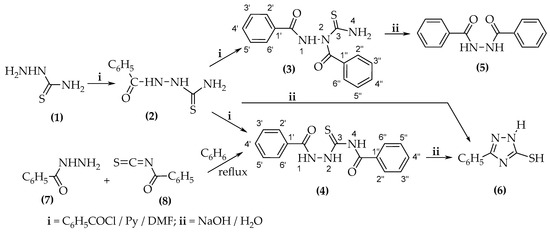
When 1-benzoylthiosemicarbazide (2) or thiosemicarbazide (1) were treated with benzoyl chloride in a basic medium, a mixture of two compounds was obtained: 1,2-dibenzoylthiosemicarbazide (3) and 1,4-dibenzoylthiosemicarbazide (4). To determine the structure of the novel compounds,
[...] Read more.
When 1-benzoylthiosemicarbazide (2) or thiosemicarbazide (1) were treated with benzoyl chloride in a basic medium, a mixture of two compounds was obtained: 1,2-dibenzoylthiosemicarbazide (3) and 1,4-dibenzoylthiosemicarbazide (4). To determine the structure of the novel compounds, 2D NMR spectroscopy techniques such as 1H-13C and 1H-15N were employed.
Full article
(This article belongs to the Collection Molecules from Side Reactions)
►▼
Show Figures
Scheme 1
Open AccessShort Note
Benzo[d][1,2,3]oxadithiole 2-Oxide
by
R. Alan Aitken, David B. Cordes, Arun Goyal and Aidan P. McKay
Molbank 2024, 2024(2), M1803; https://doi.org/10.3390/M1803 - 7 Apr 2024
Abstract
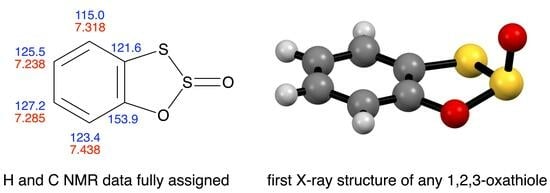
A simplified synthesis of the title compound is reported and its 1H and 13C NMR data are fully assigned including determination of H–H and C–H coupling constants. Its X-ray structure has been determined for the first time. NMR data are also
[...] Read more.
A simplified synthesis of the title compound is reported and its 1H and 13C NMR data are fully assigned including determination of H–H and C–H coupling constants. Its X-ray structure has been determined for the first time. NMR data are also presented for the oxygen analogue.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures
Graphical abstract
Open AccessShort Note
(2R,4aS,6aS,12bR,14aS,14bR)-N-(2-(2-(2-(2-Azidoethoxy)ethoxy)ethoxy)ethyl)-10-hydroxy-2,4a,6a,9,12b,14a-hexamethyl-11-oxo-1,2,3,4,4a,5,6,6a,11,12b,13,14,14a,14b-tetradecahydropicene-2-carboxamide
by
Guo Yuzhu, Margrate Anyanwu, Xiao Yang, Ren Zimo, Alessandra Gianoncelli, Giovanni Ribaudo and Paolo Coghi
Molbank 2024, 2024(2), M1800; https://doi.org/10.3390/M1800 - 1 Apr 2024
Abstract
►▼
Show Figures
In this report, we discuss the synthesis of a compound obtained from the derivatization of the natural compound celastrol. This derivative is connected to PEG azide moiety through an amide linkage. The linkage was achieved through the activation of the carboxylic acid using
[...] Read more.
In this report, we discuss the synthesis of a compound obtained from the derivatization of the natural compound celastrol. This derivative is connected to PEG azide moiety through an amide linkage. The linkage was achieved through the activation of the carboxylic acid using HOBt/EDC. The compound was fully characterized by proton (1H), carbon-13 (13C), heteronuclear single quantum coherence (HSQC), correlation spectroscopy (1H-1H-COSY), and distortionless enhancement by polarization transfer (DEPT) NMR. Ultraviolet (UV), Fourier-transform infrared (FTIR), and high-resolution mass spectrometry (HRMS) were also adopted. Computational investigations were conducted to forecast the binding mode between the synthesized compound and sarco-endoplasmic reticulum (SR) Ca2+ transport ATPase (SERCA), a known target for the development of novel therapeutics for rheumatoid arthritis. Additionally, the drug-likeness of the synthesized compound was assessed by predicting its pharmacokinetic properties.
Full article
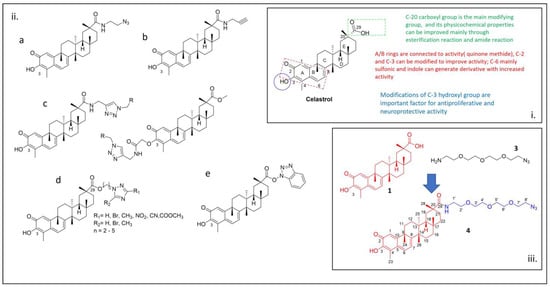
Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Catalysts, Chemistry, Materials, Molbank, Molecules
Catalysis: Homogeneous and Heterogeneous, 2nd Edition
Topic Editors: Luísa Margarida Martins, Isidro M. PastorDeadline: 31 October 2024
Topic in
Catalysts, Chemistry, Inorganics, Molbank, Molecules, Polymers
Heterocyclic Carbene Catalysis
Topic Editors: Sabine Berteina-Raboin, Thierry Besson, Patrick RollinDeadline: 30 November 2024
Topic in
Catalysts, Chemistry, Molbank, Molecules, Sustainable Chemistry
Towards the Sustainable Synthesis of Biologically Active Molecules in Green Solvents
Topic Editors: Antonio Salomone, Serena PerroneDeadline: 31 December 2024

Conferences
Special Issues
Topical Collections
Topical Collection in
Molbank
Molecules from Catalytic Processes
Collection Editor: Nicola Della Ca’


![Chlorido-pentamethylcyclopentadienyl-[2-(2-pyridyl-к<em>N</em>)-ferrocenyl-к<em>C</em>]-iridium(III)](https://pub.mdpi-res.com/title_story/title_story_17142961226993.jpg?1715181861)
![4,4′-([2,2′-Bithiophene]-5,5′-diylbis(ethyne-2,1-diyl))bis(1-methylpyridin-1-ium) Iodide](https://pub.mdpi-res.com/title_story/title_story_17109284201736.png?1715181861)




